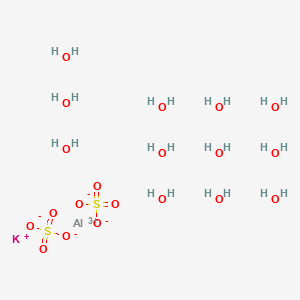
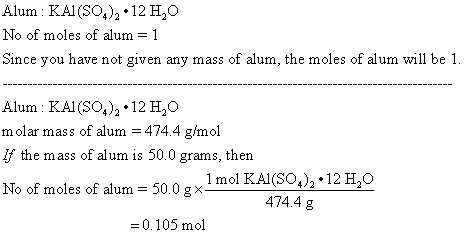Cubic structure of alum K(Al,Cr)(SO 4 ) 2 ⋅12H 2 O, space group Pa3 _ .... | Download Scientific Diagram

PPT - Exp 12 Synthesis: Preparation of Alum Alum: potassium aluminum sulfate dodecahydrate PowerPoint Presentation - ID:5409973
✓ Solved: A sample of potassium aluminum sulfate 12-hydrate, KAl(SO4)2·12H2O, containing 101.5 mg is...

SOLVED: There are several steps to the synthesis of the aluminum potassium alum that can be represented by the overall reaction.2Al(s) + 2KOH(aq) + 4H2SO4(aq) + 24H2O(l) → 2KAl(SO4)212H2O(s) + 3H2(g) +H2O(l)1.

Kal(so4)2 12h2o Aluminum Potassium Sulfate Lump Potassium Alum Potash Alum - Buy Potash Alum,Aluminum Potassium Sulfate,Kal(so4)2 12h2o Potash Alum Product on Alibaba.com

Kal(so4)2 12h2o Potash Alum Potassium Alum Stone - Buy Potassium Alum Stone,Potash Alum Stone,Kal(so4)2 12h2o Potassium Alum Stone Product on Alibaba.com
Cubic structure of alum K(Al,Cr)(SO 4 ) 2 ⋅12H 2 O, space group Pa3 _ .... | Download Scientific Diagram

High Quality Potash Alum Kal(so4)2.12h2o - Buy High Purity Aluminium Potassium Sulfate,Aluminium Potassium Sulphate For Food Addictive,Food Grade Aluminium Potassium Sulphate Product on Alibaba.com

Suppose Harry begins with the hydrate KAl(SO4)2·12H2O. After dehydration he finds that he is left with 3.0 - Brainly.com

SOLVED: KAl(SO4)2 reacts with BaCl2 in aqueous solutions to produce a precipitate of BaSO4 that can be filtered and weighed. The balanced equation for the reaction is: KAl(SO4)2 (aq) + 2 BaCl2 (

Anomalous longitudinal acoustic phonon and elastic constant in potassium alum KAl(SO4)2·12H2O single crystal studied by Brillouin spectroscopy - ScienceDirect